TAMBE at Geisinger: Reducing risk, improving outcomes
The TAMBE procedure is light years ahead of traditional surgery for thoracoabdominal aortic aneurysms — especially for patients at high risk for complications.
Geisinger has long been a leader in providing minimally invasive surgery for complex aortic aneurysms in northeastern and central Pennsylvania. And now, TAMBE has been improved thanks to FDA approval of a device specifically designed for high-risk patients.
Traditionally, surgery for complex aortic aneurysms has involved an incision from the shoulder blade to the navel, says Geisinger vascular surgeon John Jeremy Irvan, MD, who has performed the TAMBE procedure for over a year, operating on more than 20 patients.
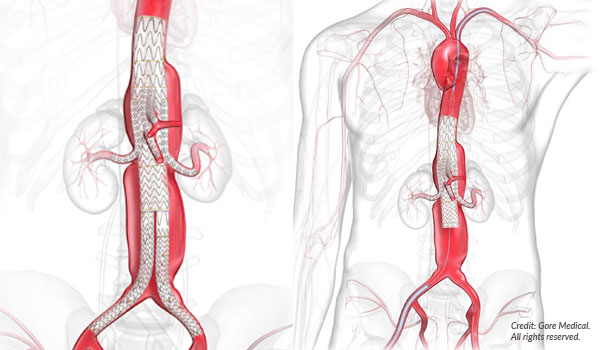
The TAMBE device is a custom-made stent graft with multiple branches designed to fit around the aneurysm. This allows blood to flow through the graft and exclude the weakened aneurysm wall.
“Implanting the device only requires a small incision in the groin,” Dr. Irvan explains. “We can avoid that majorly invasive surgery.”
Dr. Irvan says he and his team were creating their own version of the TAMBE device on site at Geisinger for several years before the commercial product became available. The FDA-approved device streamlines the process and makes it available to more patients.
“We can now offer this life-saving procedure to people who otherwise couldn’t have surgery due to the potential for complications,” he says. “This gives us something premade and purposely built.”
Most importantly, patients in Geisinger’s service area no longer have to travel long distances for this type of lower-risk surgery. “We’re offering advanced care locally,” Dr. Irvan explains. “We have good outcomes — patients are doing well, right here.”